| 人iPS细胞 | ||||||
| 品牌 | Code No. | 产品名称 | 包装量 | 价格(元) | 说明书 | 数量 |
| Cellartis | Y00185 | Cellartis® human iPS cell line P11025 Kit | 1 Kit | ¥23,632 | |
  |
| Cellartis | Y00275 | Cellartis® human iPS cell line 7 (ChiPSC7) Kit | 1 Kit | ¥22,525 | |
  |
| Cellartis | Y00285 | Cellartis® human iPS cell line 12 (ChiPSC12) Kit | 1 kit | ¥22,525 | |
  |
| Cellartis | Y00315 | Cellartis® human iPS cell line 21 (ChiPSC21) Kit | 1 Kit | ¥22,525 | |
  |
| Cellartis | Y00325 | Cellartis® human iPS cell line 22 (ChiPSC22) Kit | 1 kit | ¥22,525 | |
  |
| Cellartis | Y00305 | Cellartis® human iPS cell line 18 (ChiPSC18) Kit | 1 Kit | ¥22,525 | |
  |
收藏产品 加入购物车
| 人iPS细胞 (Human Induced Pluripotent Stem Cells) |
||||||
|
||||||
| ■ 关联产品 | ||||||
|
||||||
| ■ 产品详情请点击: |
||||||
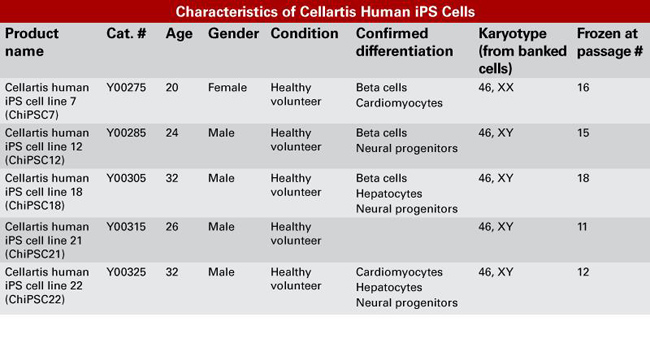 |
||||||
| Characteristics of Cellartis human iPS cells. The cell lines were generated from fibroblasts using defective polycistronic retrovirus technology to deliver Oct-4, Sox-2, KLF-4, and c-Myc. The skin fibroblasts were obtained from donors of varying age and gender, have a normal karyotype, and have been confirmed to differentiate into specific cell lineages. Consent was obtained from all donors for commercial use. | ||||||
| 参考文献: | ||||||
| 1. de Peppo GM, et al. Osteogenic potential of human mesenchymal stem cells and human embryonic stem cell-derived mesodermal progenitors: a tissue engineering perspective. Tissue Eng Part A.2010 Nov;16(11):3413-3426. 2. Both SK, et al. Differential bone-forming capacity of osteogenic cells from either embryonic stem cells or bone marrow-derived mesenchymal stem cells. J Tissue Eng Regen Med.2011 Mar;5(3):180-190. 3. de Peppo GM, et al. Free-form-fabricated commercially pure Ti and Ti6A14V Porous Scaffolds support the growth of human embryonic stem cell-derived mesodermal progenitors. ScientificWorldJournal. 2012;2012:646417.Epub 2012 Jan 4. 4. Ou Li, et al. Human embryonic stem cell-derived mesenchymal stroma cells(hES-MSCs) engraft in vivo and support hematopoiesis without suppression immune function: implications for off –the shelf ES-MSC therapies. PLOS ONE.2013; 8(1):e55319. |
||||||
